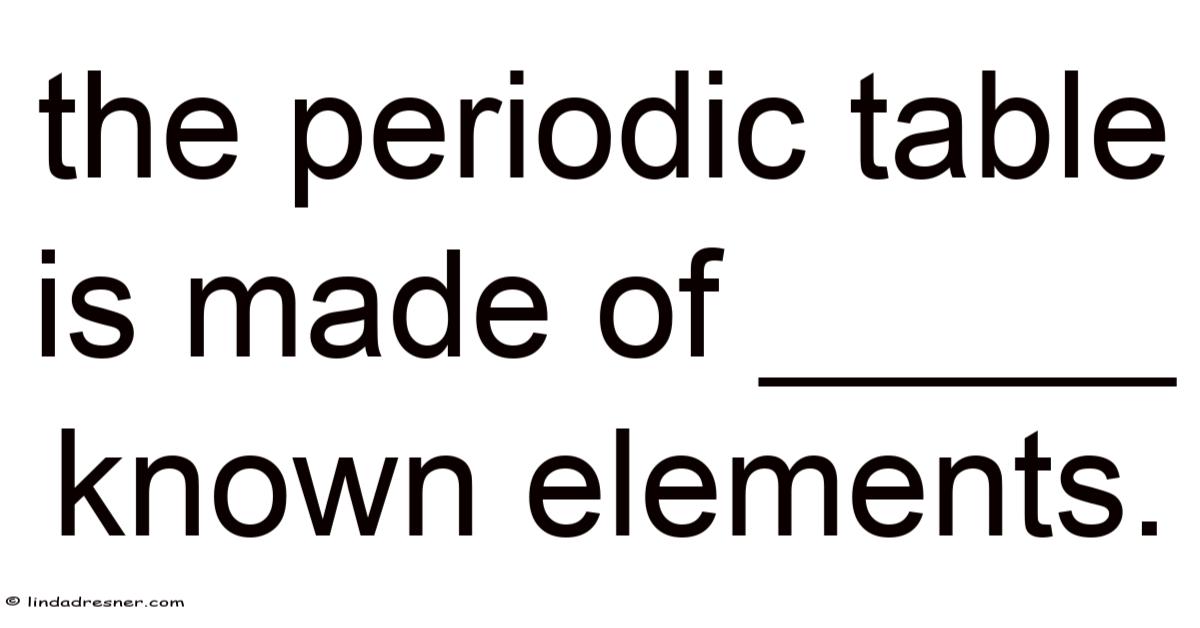The Periodic Table is Made of 118 Known Elements
The periodic table, a cornerstone of modern chemistry, organizes the fundamental building blocks of matter—elements—into a structured framework that reveals patterns in their properties and behaviors. As of now, the periodic table contains 118 known elements, each uniquely identified by its atomic number, which corresponds to the number of protons in its nucleus. Still, these elements range from naturally occurring substances like hydrogen and oxygen to synthetic creations such as oganesson, the heaviest element currently recognized. This article explores the composition of the periodic table, the discovery of its elements, and the scientific principles that govern their arrangement.
The Structure of the Periodic Table
The periodic table is divided into rows called periods and columns known as groups or families. Elements in the same group share similar chemical properties due to their identical valence electron configurations. As an example, alkali metals in Group 1 (e.g., lithium, sodium) are highly reactive, while noble gases in Group 18 (e.g., helium, neon) are inert. The table also categorizes elements into metals, nonmetals, and metalloids, each with distinct physical and chemical traits. Metals, which dominate the left side of the table, are typically shiny, malleable, and good conductors of heat and electricity. Nonmetals, found on the right, include gases like nitrogen and solids like carbon, while metalloids, such as silicon, exhibit properties of both.
The Discovery of Elements
The periodic table’s evolution reflects humanity’s quest to understand matter. The first elements, such as gold, silver, and lead, were known to ancient civilizations, but systematic discovery began in the 18th century. In 1789, French chemist Antoine Lavoisier compiled a list of 33 elements, marking the start of modern chemistry. The 19th century saw breakthroughs like Dmitri Mendeleev’s 1869 periodic table, which arranged elements by atomic weight and predicted the existence of undiscovered elements, such as gallium and germanium. Mendeleev’s work laid the foundation for the modern table, which now includes 118 elements.
Natural vs. Synthetic Elements
Of the 118 elements, 94 occur naturally on Earth, while the remaining 24 are synthetic, created in laboratories through nuclear reactions. Natural elements include hydrogen, helium, and carbon, which form the basis of life and industry. Synthetic elements, such as technetium (element 43) and plutonium (element 94), are produced in particle accelerators or nuclear reactors. These elements are often unstable and decay rapidly, but their study provides insights into nuclear physics and the limits of atomic structure Nothing fancy..
The Role of Atomic Number and Isotopes
Each element is defined by its atomic number, the count of protons in its nucleus. As an example, carbon has an atomic number of 6, meaning it has six protons. While the number of neutrons can vary, creating isotopes (e.g., carbon-12 and carbon-14), the atomic number remains constant for a given element. This distinction is crucial for understanding chemical behavior, as isotopes of the same element share similar properties but differ in mass Small thing, real impact. Simple as that..
The Future of the Periodic Table
Scientists continue to push the boundaries of the periodic table. Elements beyond 118, such as ununpentium (element 115) and ununhexium (element 116), have been synthesized in recent years, though their stability remains a challenge. These "superheavy" elements exist for only fractions of a second, but their discovery expands our understanding of nuclear forces and the periodic table’s limits Worth keeping that in mind. Worth knowing..
Conclusion
The periodic table’s 118 elements represent the culmination of centuries of scientific exploration. From the ancient use of metals to the creation of synthetic superheavy elements, each discovery has deepened our understanding of the universe. As research advances, the table may one day include even more elements, continuing to shape the fields of chemistry, physics, and materials science. The periodic table is not just a chart—it is a dynamic tool that reflects the ever-evolving nature of scientific knowledge That alone is useful..
FAQs
Q: How many elements are in the periodic table?
A: The periodic table currently contains 118 known elements, with 94 occurring naturally and 24 synthesized in laboratories.
Q: What is the difference between natural and synthetic elements?
A: Natural elements occur in nature, while synthetic elements are created artificially through nuclear reactions.
Q: Why are some elements unstable?
A: Elements with high atomic numbers, particularly those beyond 83 (bismuth), are often unstable due to the imbalance between protons and neutrons in their nuclei, leading to rapid decay No workaround needed..
Q: How are new elements discovered?
A: New elements are typically created in particle accelerators by colliding atomic nuclei, a process that requires advanced technology and precise control The details matter here..
Q: What is the significance of the periodic table’s structure?
A: The periodic table’s arrangement reveals patterns in element properties, allowing scientists to predict behaviors and discover new elements based on periodic trends.
Understanding persists as a bridge between past and future discoveries.
Conclusion
The periodic table remains a cornerstone, guiding explorers through its detailed web Practical, not theoretical..
The periodic table is not just a chart—it is
a living document that evolves with each new discovery. Its structure not only catalogs the building blocks of matter but also serves as a roadmap for innovation. In modern times, the periodic table underpins advancements in technology, from the rare earth elements powering smartphones to the semiconductors enabling quantum computing. Researchers are already exploring how superheavy elements might exhibit unique properties, such as enhanced stability or novel chemical behaviors, which could revolutionize fields like energy storage or medical imaging.
Beyond the lab, the periodic table shapes education and culture. Also, it is a universal language for scientists, a symbol of human curiosity, and a reminder of our interconnectedness with the cosmos. As we uncover more about the universe’s fundamental components, the table will continue to adapt, reflecting the infinite complexity of the matter that surrounds us Less friction, more output..
This is the bit that actually matters in practice.
Final Thoughts
The periodic table is more than a static chart—it is a testament to humanity’s relentless pursuit of knowledge. Each element tells a story of cosmic origins, human ingenuity, and the delicate balance of forces that govern our existence. Whether unlocking the secrets of superheavy atoms or harnessing the potential of familiar elements, the table remains an enduring beacon of discovery, guiding us toward a future where the boundaries of science are constantly redefined.
Recent breakthroughs hint at untapped potential within its boundaries Worth keeping that in mind..
Conclusion
The periodic table endures as a testament to humanity’s quest for understanding, bridging epochs through its enduring relevance That's the part that actually makes a difference..
The quest to understand the universe continues to unfold, with each new element adding another layer to our comprehension of matter’s fundamental nature. As scientists push the limits of discovery, the periodic table remains an essential tool, not only for identifying elements but also for inspiring future innovations.
This progression underscores the importance of curiosity and perseverance, reminding us that every atom carries within it a piece of the cosmos. By bridging the gap between theory and experiment, the periodic table fosters a deeper appreciation for the structure of reality Practical, not theoretical..
In this journey, the elements beyond bismuth symbolize both challenges and opportunities. Their study encourages us to think beyond the present, envisioning how tomorrow’s discoveries might reshape our relationship with the material world.
Conclusion
The periodic table stands as a dynamic testament to human intellect, a guide that connects our past achievements to the possibilities of the future. Its evolution reflects our enduring drive to explore, innovate, and redefine the limits of what we know Easy to understand, harder to ignore..