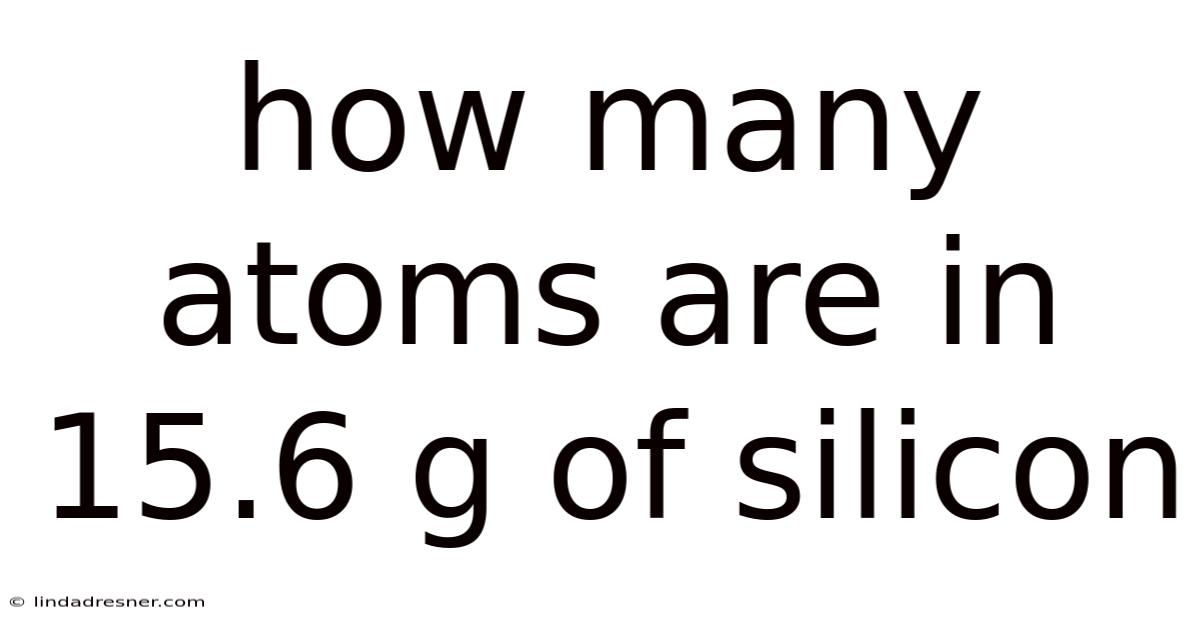The layered dance of atoms underpins the very fabric of the physical universe, shaping everything from the microscopic structures of matter to the macroscopic properties that define our world. Yet, determining precisely how many atoms reside within a given mass of silicon requires a meticulous approach that bridges chemistry, physics, and mathematics. Because of that, its atomic structure, composed predominantly of silicon atoms interwoven with oxygen, forms the foundation of countless materials, from glass to semiconductors. So naturally, by unraveling the complexities behind atomic arrangements, we gain insight into the material properties that influence technological advancements, environmental sustainability, and human innovation. On the flip side, at the core of this understanding lies the concept of atomic composition, a principle that governs how elements interact, combine, and transform under varying conditions. This article digs into the scientific process of calculating atomic counts, offering readers a comprehensive exploration of the principles involved while emphasizing the practical implications of such knowledge. Which means silicon, a silicate mineral renowned for its prevalence in technology and construction, exemplifies this universal truth. The journey begins with grasping the fundamental concepts that enable precise measurements and informed decision-making, setting the stage for a deeper appreciation of the microscopic world that shapes our daily lives.
Understanding Atomic Mass and Molar Mass
At the heart of any atomic calculation lies the distinction between atomic mass and molar mass, two terms that often confuse learners but are crucial for accurate results. Atomic mass, a measure of the average mass of a single atom of an element in atomic mass units (amu), provides a snapshot of an element’s composition, while molar mass, expressed in kilograms per mole, represents the total mass required to contain one mole of a substance. These two quantities are not merely numerical values but serve as bridges between the atomic scale and the macroscopic world. For silicon (Si), whose atomic mass is approximately 28.085 g/mol, understanding its molar mass becomes essential. Converting grams to moles necessitates dividing the given mass by the molar mass, a process that demands precision. Consider a scenario where 15.6 grams of silicon is taken as a starting point. Here, dividing 15.6 g by 28.085 g/mol yields approximately 0.558 moles. This foundational step underscores the importance of accurate calculations, as even minor errors can cascade into significant discrepancies when scaling up to larger quantities. The interplay between atomic mass and molar mass thus forms the bedrock of quantitative analysis, enabling scientists and engineers to translate theoretical knowledge into tangible applications And that's really what it comes down to. That alone is useful..
The Role of Moles in Atomic Counting
Moles act as a key intermediary between the microscopic world of atoms and the measurable quantities we observe in labs and industries. By defining one mole as precisely 6.022 × 10²³ particles—Avogadro’s number—the concept of atomic counting becomes accessible. This number, often denoted as Avogadro’s constant, serves as a universal constant bridging the gap between individual atoms and collective entities. For silicon, applying this principle means converting grams into moles and subsequently into atoms. The process involves not only calculating moles but also recognizing that each mole contains an exact number of particles, ensuring consistency across different measurement scales. Here's a good example: if 0.558 moles of silicon are present, multiplying by Avogadro’s number reveals the exact count: 0.558 × 6.022 × 10²³ ≈ 3.35 × 10²³ atoms. Such precision is vital in fields ranging from pharmaceuticals, where dosage accuracy hinges on precise atomic-level control, to materials science, where the arrangement of atoms dictates a material’s strength or conductivity. The molar framework thus acts as a scaffold, allowing practitioners to visualize how atomic arrangements translate into observable outcomes.
From Moles to Atoms: The Mathematical Foundation
The transition from moles to individual atoms requires careful application of mathematical principles, where precision is essential. Starting with the moles calculated earlier—0.558 moles—one must multiply by Avogadro’s number to attain atomic quantities. Even so, this process is not without its challenges. Ensuring that unit conversions
remain consistent is critical; the mass in grams must align with the molar mass, and the resulting moles must be handled with exactitude. 558 moles by 6.For silicon, the multiplication of 0.The calculation, while straightforward in theory, demands rigorous attention to detail. 022 × 10²³ atoms/mol results in a staggeringly large number, reinforcing the sheer scale of atomic interactions. Any rounding during intermediate steps can distort the final atom count, particularly in high-stakes environments like semiconductor manufacturing, where even nanometer-scale deviations can alter device performance. This mathematical journey highlights the elegance of stoichiometry—it transforms abstract masses into concrete, countable entities, providing a clear window into the composition of matter Still holds up..
Practical Implications and Conclusion
The ability to convert between mass, moles, and atoms is not merely an academic exercise but a fundamental skill with far-reaching implications. In fields such as chemistry, engineering, and materials science, these conversions enable the design of alloys, the optimization of chemical reactions, and the development of nanotechnologies. For silicon, a cornerstone of the semiconductor industry, precise molar and atomic calculations ensure the reliability and efficiency of electronic components, from microprocessors to solar cells. On top of that, this understanding fosters a deeper appreciation for the interconnectedness of the microscopic and macroscopic worlds, where abstract numbers translate into real-world properties. In the long run, mastering the conversion of grams to moles and atoms equips professionals with the tools to manipulate matter at its most fundamental level, driving innovation and precision across scientific and industrial domains.
Beyond the Calculation: A Foundation for Innovation
The seemingly simple conversion between grams, moles, and atoms unlocks a deeper understanding of the building blocks of our universe. It’s a cornerstone of scientific inquiry, allowing us to predict and control the behavior of matter with increasing accuracy. This foundational skill extends far beyond the laboratory, influencing everything from the development of new pharmaceuticals with tailored molecular structures to the creation of advanced materials with unprecedented properties.
Consider the burgeoning field of nanotechnology. That said, the precise manipulation of individual atoms and molecules is essential for building nanoscale devices, such as quantum dots and carbon nanotubes, which hold immense promise for applications in computing, medicine, and energy storage. Without a solid grasp of stoichiometry, designing and fabricating these complex structures would be virtually impossible. Similarly, in environmental science, understanding the molar and atomic composition of pollutants is vital for developing effective remediation strategies Simple, but easy to overlook. Which is the point..
Also worth noting, the principles underpinning these conversions are integral to ensuring safety and quality control in various industries. In the food industry, for example, precise calculations are used to determine nutrient content and ensure adherence to labeling regulations. In the pharmaceutical industry, accurate stoichiometry is critical for drug formulation and dosage control Worth keeping that in mind..
So, to summarize, the conversion of grams to moles and atoms is far more than a mathematical exercise. It represents a fundamental bridge between the abstract world of numbers and the tangible reality of matter. Day to day, it’s a skill that empowers scientists, engineers, and professionals across a wide range of disciplines to manipulate, understand, and innovate with matter at its most fundamental level. This seemingly simple concept underpins countless advancements and continues to drive progress in our increasingly complex world, solidifying its place as a critical tool for scientific discovery and technological innovation Surprisingly effective..
The mastery of converting grams to moles and atoms is not merely an academic exercise—it is a vital skill that shapes the trajectory of scientific and technological progress. By bridging the gap between theoretical models and tangible outcomes, this process empowers researchers and engineers to design, test, and refine solutions with remarkable precision. As industries continue to push the boundaries of what is possible, the ability to work through these conversions becomes an essential asset, fostering innovation across fields Turns out it matters..
Looking ahead, the integration of this knowledge will play a central role in addressing global challenges, from sustainable energy solutions to advanced medical technologies. Each calculation reinforces a deeper appreciation for the harmony between the microscopic and the macroscopic, reinforcing the idea that understanding matter at its core unlocks transformative possibilities.
In essence, this skill is more than numbers on a page—it is a catalyst for discovery, a foundation for precision, and a testament to human ingenuity. Embracing it ensures that professionals remain equipped to shape the future with confidence and clarity Surprisingly effective..
Conclusion: The seamless mastery of conversions between grams, moles, and atoms is a cornerstone of scientific advancement. It not only enhances our understanding of the material world but also drives the innovations that define modern progress.