_______ Are Different Forms Of A Single Element.
lindadresner
Mar 12, 2026 · 4 min read
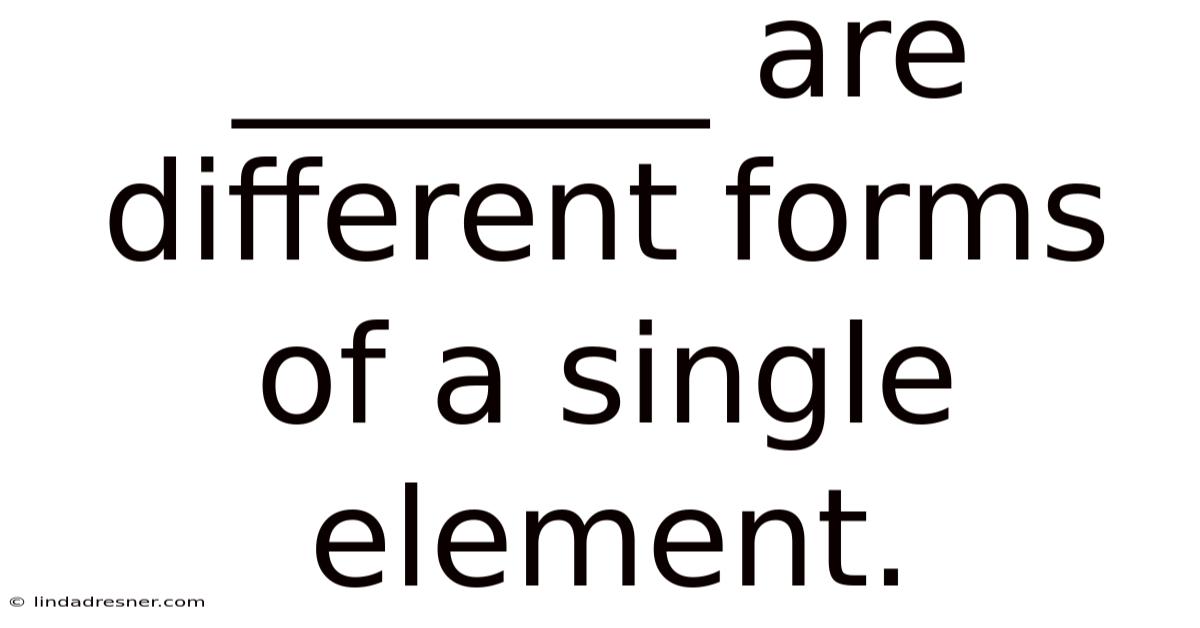
Table of Contents
Understanding Allotropes: Different Forms of a Single Element
When we think of elements, we often imagine them as fixed entities with specific properties. However, nature is full of surprises, and many elements can exist in multiple forms, each with distinct characteristics. These varying forms of a single element are called allotropes. The term “allotrope” comes from the Greek word allos, meaning “other,” and it highlights the fascinating diversity that can arise from a single type of atom. This article explores what allotropes are, why they exist, and how they impact our world.
What Are Allotropes?
At their core, allotropes are different structural forms of the same element. While the chemical composition remains unchanged, the arrangement of atoms differs, leading to variations in physical and chemical properties. For example, carbon can exist as diamond, graphite, or graphene—each with unique hardness, conductivity, and appearance. This phenomenon is not limited to carbon; other elements like oxygen, sulfur, and phosphorus also exhibit allotropy.
The existence of allotropes challenges the notion that an element’s properties are static. Instead, it reveals how subtle changes in atomic bonding or molecular structure can create entirely new materials. These differences arise from the way atoms bond with each other, influenced by factors like temperature, pressure, and the number of valence electrons.
Examples of Allotropes in Nature
To grasp the concept of allotropes, let’s examine some well-known examples. These cases illustrate how a single element can manifest in multiple forms, each with its own set of characteristics.
Carbon: The Versatile Element
Carbon is perhaps the most famous element for allotropy. Its allotropes include:
- Diamond: A crystalline structure where each carbon atom is bonded to four others in a tetrahedral arrangement. This results in extreme hardness and a sparkling appearance.
- Graphite: Here, carbon atoms are arranged in flat layers held together by weak van der Waals forces. This structure makes graphite soft and slippery, ideal for pencils.
- Fullerene (C60): A spherical molecule resembling a soccer ball, discovered in 1985. Fullerenes have unique electronic properties and are used in nanotechnology.
- Graphene: A single layer of graphite, known for its exceptional strength and conductivity.
Each of these forms serves different purposes. Diamonds are used in jewelry, graphite in writing tools, and graphene in advanced electronics.
Oxygen: Breathable and Ozone
Oxygen exists in two primary allotropic forms:
- O₂ (dioxygen): The molecule we breathe, essential for cellular respiration.
- O₃ (ozone): A triatomic molecule formed when oxygen molecules are split by ultraviolet light. Ozone is crucial for absorbing harmful UV radiation in the Earth’s stratosphere.
While both are vital, their roles and properties differ significantly. O₂ is stable
and supports life, while O₃ is highly reactive and acts as a protective shield.
Sulfur: Rings and Chains
Sulfur exhibits several allotropes, including:
- Rhombic Sulfur: A yellow, crystalline form stable at room temperature.
- Monoclinic Sulfur: Another yellow crystalline form, often formed when rhombic sulfur is heated and cooled.
- Plastic Sulfur: A black, amorphous form created by rapidly cooling molten sulfur. It has a rubbery texture and can be molded.
These forms differ in their crystal structure and stability, influencing their reactivity and applications.
Phosphorus: White, Red, and Black
Phosphorus presents a fascinating array of allotropes, each with distinct properties and hazards:
- White Phosphorus: A highly reactive, toxic, and flammable solid that glows in the dark. It spontaneously ignites in air.
- Red Phosphorus: A more stable, less reactive form used in matches and fertilizers.
- Black Phosphorus: The most thermodynamically stable form, exhibiting semiconductor properties and potential applications in electronics.
The Impact of Allotropy on Technology and Industry
The existence of allotropes has profoundly impacted various technological fields. Understanding and manipulating these different forms allows scientists and engineers to tailor materials for specific applications.
- Materials Science: Allotropy is a cornerstone of materials science. Researchers actively explore new allotropes and ways to control their formation, leading to the development of stronger, lighter, and more conductive materials. Graphene, for instance, has spurred immense research into flexible electronics, energy storage, and composite materials.
- Electronics: The unique electrical properties of allotropes like graphene and black phosphorus are revolutionizing electronics. These materials offer the potential for faster, more efficient, and flexible electronic devices.
- Energy Storage: Allotropes are being investigated for use in batteries and supercapacitors. Their high surface area and unique electronic properties can enhance energy storage capacity and charging rates.
- Medicine: Fullerenes and other allotropes are being explored for drug delivery, medical imaging, and even as potential therapeutic agents. Their ability to encapsulate molecules and target specific cells holds great promise.
- Industrial Processes: The different forms of sulfur and phosphorus are crucial in various industrial processes, from fertilizer production to the manufacture of matches and chemicals.
Conclusion
Allotropy is a fundamental concept in chemistry that highlights the remarkable versatility of elements. It demonstrates that the arrangement of atoms, rather than just the type of atom, dictates a material’s properties. From the brilliance of diamonds to the protective layer of ozone, allotropes play a vital role in our world. As research continues to uncover new allotropes and refine our understanding of their behavior, we can anticipate even more groundbreaking applications that will shape the future of technology, medicine, and industry. The study of allotropy is not just about understanding the building blocks of matter; it’s about unlocking the potential to create materials with unprecedented properties and capabilities.
Latest Posts
Latest Posts
-
In A Study Of Retractions In Biomedical Journals
Mar 12, 2026
-
Theme Statement From Romeo And Juliet
Mar 12, 2026
-
Rn Community Health Online Practice 2023 B
Mar 12, 2026
-
Unit 2 Progress Check Mcq Part A
Mar 12, 2026
-
Nih Stroke Scale Answers Group A
Mar 12, 2026
Related Post
Thank you for visiting our website which covers about _______ Are Different Forms Of A Single Element. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.